Scientists have meticulously charted the olfactory landscape of the mouse nose, a groundbreaking achievement that has overturned long-held assumptions about how this fundamental sense operates. The first-ever comprehensive map of smell receptors reveals a sophisticated, highly organized system, challenging the previous notion of random distribution within the nasal cavity. This discovery not only deepens our understanding of olfaction in mice but also holds significant implications for deciphering the complexities of smell in humans and potentially developing new therapeutic strategies for olfactory disorders.
The Genesis of Olfactory Mapping: A Technological Leap
For decades, the prevailing scientific consensus suggested that the approximately 1,172 different types of olfactory receptors, each capable of detecting a distinct smell, were scattered haphazardly throughout the lining of the nasal cavity. This stood in stark contrast to other sensory systems, such as vision, hearing, and touch, which are known to employ precise spatial maps. In the auditory system, for instance, different sound frequencies are processed at specific locations within the cochlea, a principle that extends to the brain’s interpretation of sound. The idea of a similar organized map for smell had remained largely elusive, hampered by technological limitations.
The paradigm shift began to emerge over the past six to seven years, driven by advancements in molecular biology and imaging techniques. Researchers gained the ability to analyze the genetic activity within individual cells, a process known as single-cell sequencing. This powerful tool allowed scientists to examine millions of neurons across hundreds of mice, identifying which specific receptor gene was active in each mature olfactory sensory neuron.
Dr. Sandeep Datta, a leading neurobiologist at Harvard Medical School and senior author of the study, explained the significance of these technological breakthroughs. "Single-cell sequencing enabled us to look at each mature olfactory sensory neuron one at a time, to identify which receptor is being expressed," Dr. Datta stated. Complementing this, a technique called spatial transcriptomics provided the crucial ability to pinpoint the precise location of these identified receptors within the nasal tissue.
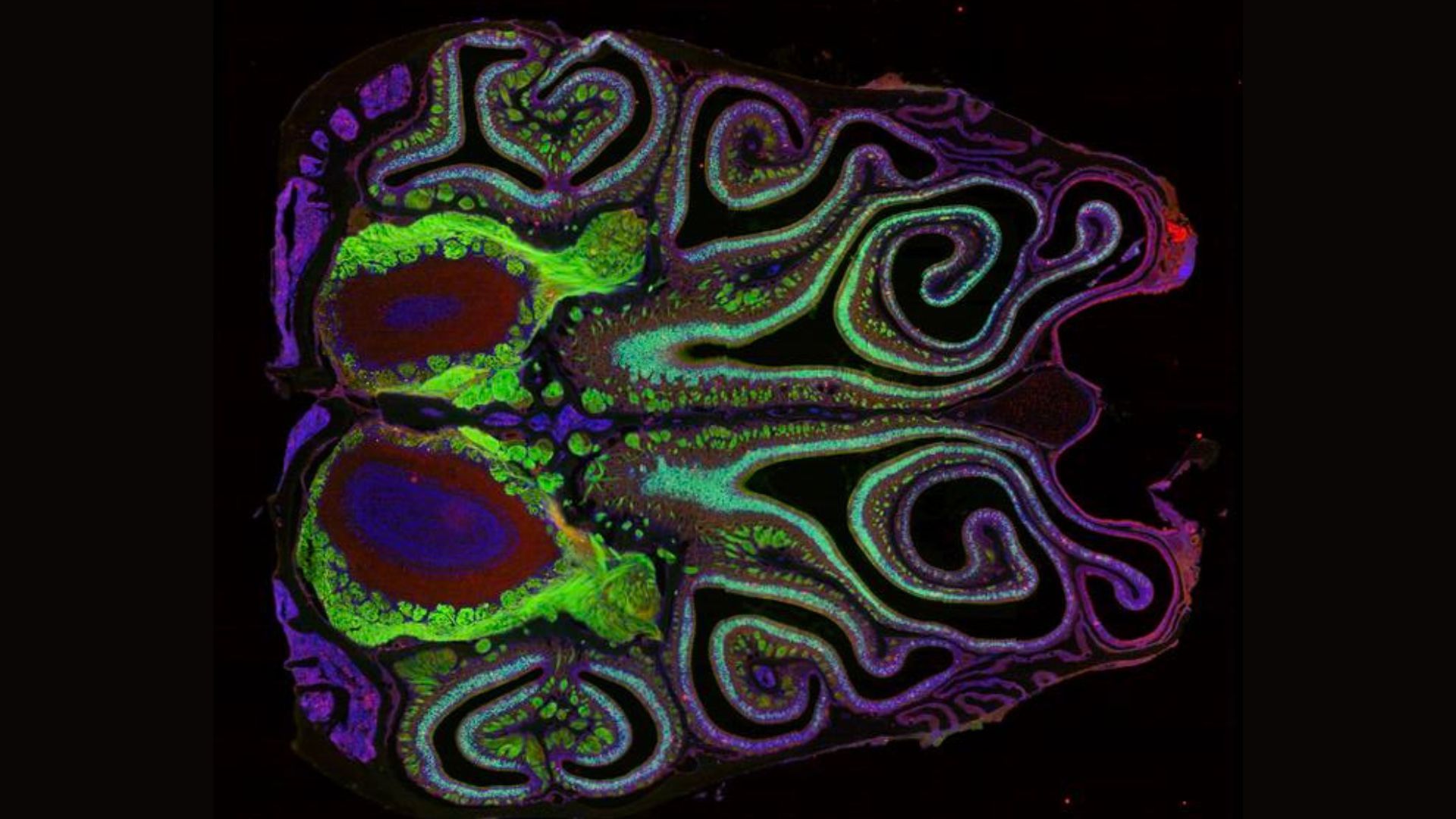
A Thousand Stripes of Scent: The Nasal Map Revealed
The culmination of these advanced techniques is a remarkably detailed map of the mouse olfactory system. The research team, leveraging data from approximately 5.5 million neurons, has unveiled an intricate arrangement of over 1,100 distinct smell receptors. "The map showed a thousand separate stripes of odor receptor expression that overlap with each other but are very organized," Dr. Datta described, characterizing the resulting visualization as a "beautiful map."
This newly constructed map vividly illustrates that olfactory receptors are not randomly distributed but are clustered into distinct, tightly packed bands within the olfactory epithelium, the specialized tissue lining the nasal cavity responsible for detecting odors. Each band is dominated by neurons expressing a particular type of olfactory receptor, creating a complex mosaic of scent detection zones.
The Brain’s Echo: A Mapped Connection
The implications of this organized nasal map extend directly to the brain. The study revealed a striking correspondence between the spatial organization of receptors in the nose and their projections in the olfactory bulb, the brain’s primary hub for processing olfactory information. Neurons that express the same receptor type in the nose converge and synapse at specific, corresponding locations within the olfactory bulb. "The map in the nose is precisely aligned with the map in the brain," Dr. Datta emphasized, highlighting a fundamental principle of neural organization. This precise alignment suggests that the brain receives olfactory information in a highly structured manner, allowing for the sophisticated interpretation of complex smells.
The sheer complexity of the mouse olfactory system is underscored by the sheer number of receptor types. Mice possess around 20 million olfactory neurons, each expressing one of over a thousand different smell receptors. This vast repertoire allows mice to perceive a far richer and more nuanced world of scents compared to humans, who have a comparatively smaller number of olfactory receptor types, estimated to be around 400.
Molecular Architects: The Role of Retinoic Acid
A key question arising from this discovery is how this intricate organization is established and maintained. The research identified a crucial molecule, retinoic acid (RA), as a likely architect of this olfactory map. RA appears to play a pivotal role in guiding each olfactory sensory neuron to express a specific receptor based on its positional information within the nasal epithelium. Experiments involving the manipulation of RA levels demonstrated a direct correlation with the shifting of receptor expression patterns, with increased RA causing upward shifts and decreased RA leading to downward shifts. This suggests that RA acts as a molecular signaling pathway, orchestrating the precise placement and functional identity of olfactory neurons.
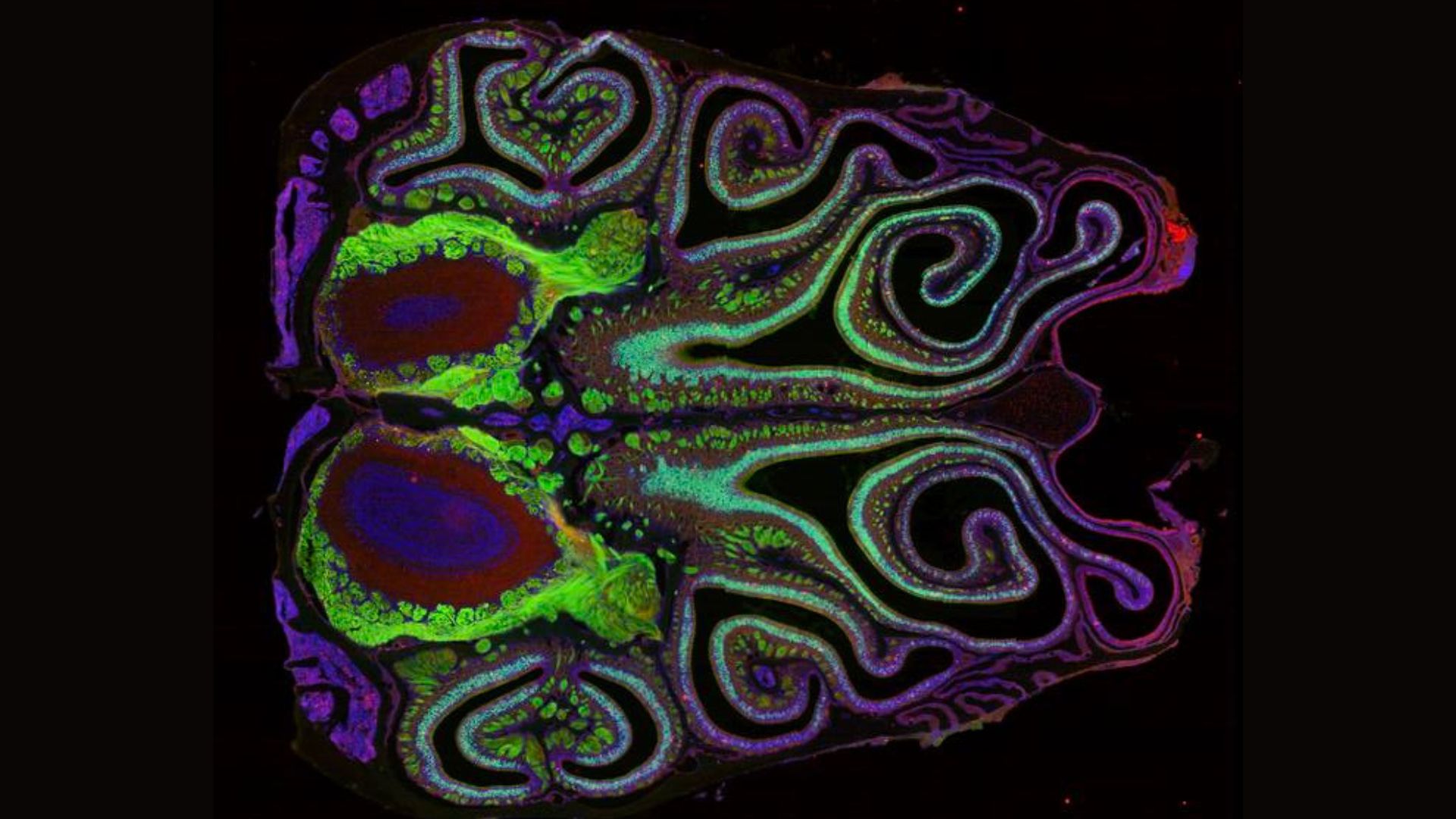
The consistency of these receptor positions across different laboratory mice further solidified the findings. The researchers observed that the approximately 1,100 types of receptors were located in essentially the same spatial arrangements in every mouse examined. This remarkable uniformity points to a conserved developmental program that ensures the robust and reproducible formation of the olfactory map.
Unraveling the "Why": Future Directions and Human Parallels
While the "how" of olfactory organization is becoming clearer, the "why" remains an area of active investigation. Scientists are now probing the evolutionary and functional advantages of such a highly ordered system. "The question we’re wondering about is, to what extent are human noses organized like this?" Dr. Datta mused, pointing towards the next frontier of olfactory research.
The human olfactory system, despite having fewer receptor types than mice, shares many fundamental similarities. Understanding whether these organizational principles observed in mice translate to humans is of paramount importance. Such knowledge could pave the way for novel therapeutic interventions for a range of olfactory disorders, including anosmia (loss of smell) and hyposmia (reduced sense of smell). These conditions can have profound impacts on quality of life, affecting everything from the enjoyment of food to the detection of danger. Furthermore, recent studies have linked a diminished sense of smell to an increased risk of depression and other neurological conditions in older adults, highlighting the broader health implications of a healthy olfactory system.
The potential for developing targeted treatments for smell loss could revolutionize the management of these debilitating conditions. By understanding the molecular mechanisms that guide receptor organization, researchers may be able to develop therapies to restore or enhance olfactory function. This could involve manipulating signaling pathways like the one involving retinoic acid or developing strategies to promote the regeneration of olfactory neurons.
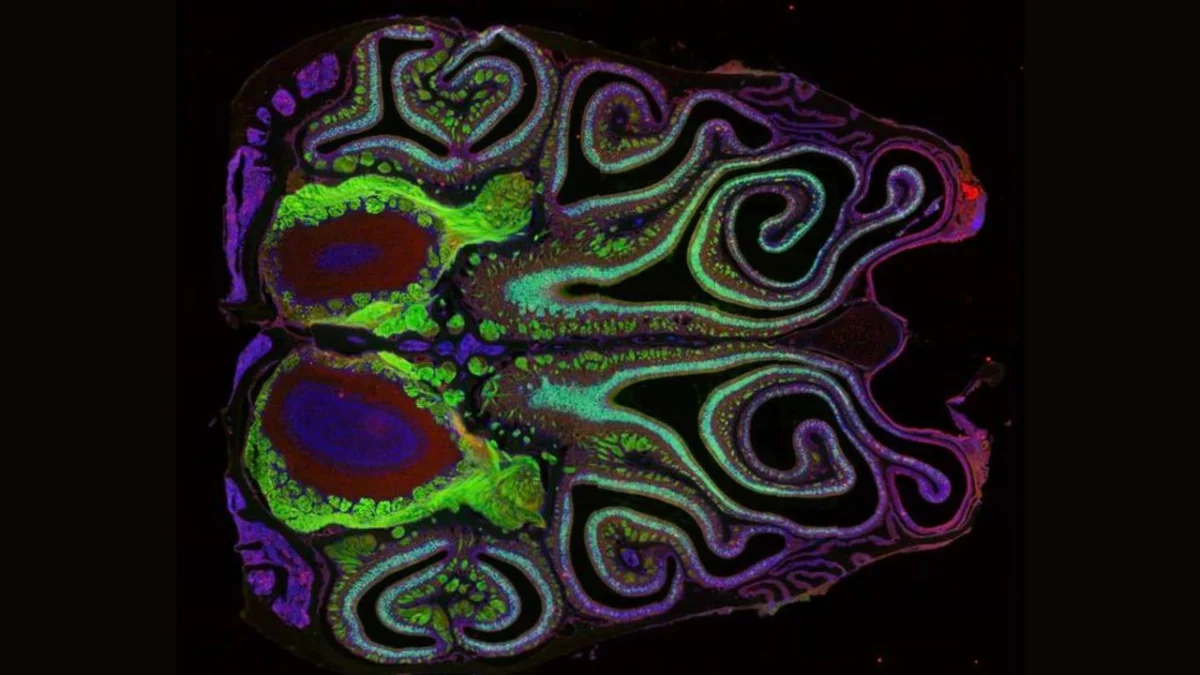
The groundbreaking work, published in a leading scientific journal, represents a significant leap forward in our understanding of one of the most fundamental and complex human senses. It not only demystifies the intricate architecture of smell in mice but also opens exciting avenues for research into human olfaction, with the ultimate goal of improving health and well-being. The detailed mapping of the olfactory system serves as a testament to the power of technological innovation in unraveling biological mysteries and underscores the intricate elegance of life’s sensory machinery. The organized stripes of scent in a mouse’s nose are, in essence, a meticulously crafted code, waiting to be fully deciphered.







Leave a Reply